
Sodium bicarbonate is, on the other hand, an sodium bicarbonate and not an alkali. 
The dough rises in this step, resulting in a chewy finish. When you add it to dough, you can use carbon dioxide and water, both of which are produced by the reaction of sugar and other ingredients. Baking soda contains no sodium bicarbonate, unlike sodium carbonate. It does not contain any gluten, which is essential for bread’s elasticity and shape. Because it is made of the same elements as table salt, it has two atoms shorter molecules. Baking Sodaĭespite this, sodium bicarbonate, unlike most other baking ingredients, is not a flour ingredient. Sodium carbonate, water, and carbon dioxide are produced by the decomposition of organic matter at temperatures greater than 50 C. Because it is so soluble in water, it has a slight alkaline flavor. A sodium bicarbonate compound is classified as an amphoteric compound because it has character acids but basic elements at the same time. NaHCO3 is a chemical formula made up of a molar mass of 84 g mol-1 and a chemical formula of NaHCO3. The Solvay method is the most commonly used method of preparation. In water, it is extremely soluble, whereas in acetone and methanol, it is extremely soluble. Salt crystals decompose at temperatures greater than 50C to form sodium carbonate. It is a white crystalline solid or fine powder with no odor or taste. The chemical formula for sodium bicarbonate is NaHCO3. It is also used as a mild alkali to neutralize acids. Sodium bicarbonate, also known as baking soda, is a white powder that is commonly used as a leavening agent in baking. This process is frequently used to produce ethanoic acid, which can be derived from other resources such as methane or ethylene. The ethanoic acid is base-catalyzed, and it is then converted to a mixture of carbon dioxide, water, and hydrogen gas. When an ethanoic acid reacts with a base or basic salt, a chemical reaction occurs. Is Nahco3 Hc2h3o2 → Nac2h3o2 H2o Co2 Balanced? What Is The Chemical Formula For Sodium Bicarbonate And Water? It is naturally soluble in water but not as completely soluble in alcohol. NaHCO3, a white powder or crystalline or granular compound with sodium bicarbonate, is also known as bicarbonate of soda or baking soda. Sodium bicarbonate has a pH of 8.5 (1% aqueous solution, 25C) and is a white powder made up of NaHCO3. 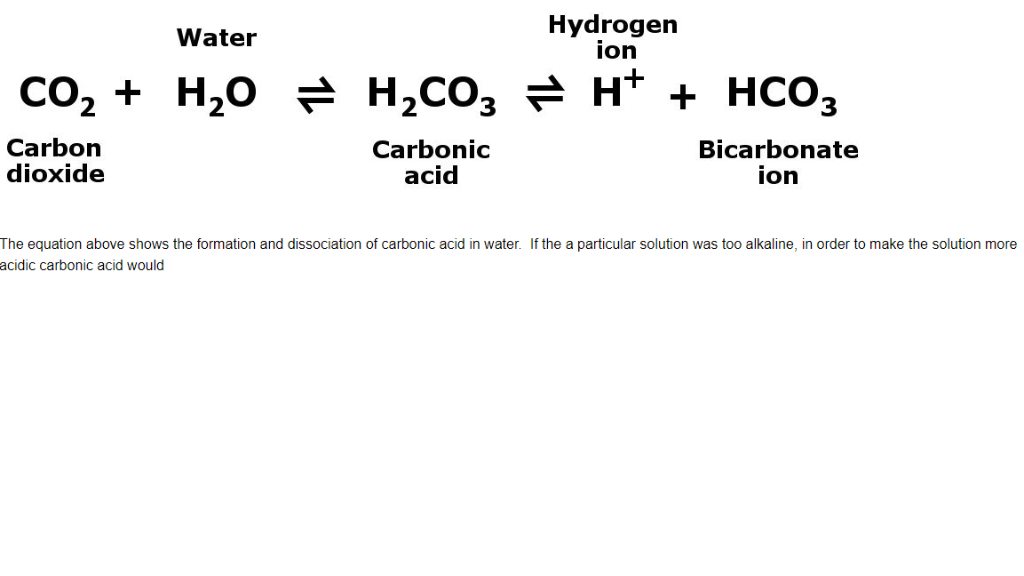
As these ions interact with acids, they create carbon dioxide gas (CO2) and water. Sodium bicarbonate, also known as baking soda or sodium hydrogen carbonate, is a white powder that readily dissolves in water and produces both sodium (Na+) and bicarbonate (HCO3) ions. Baking soda can be used as a natural pesticide in the garden to kill harmful insects and fungi. When baking powder is combined with water or milk, alkali and acid react to produce carbon dioxide bubbles. The reason for this is that baking soda is an alkali, which is a chemical that is the opposite of acid. This process involves combining concentrated brine with ammonia and carbon dioxide, which are then mixed. Nacolite, a mineral, contains sodium hydrogen carbonate. Many of its claims do not have solid scientific backing. Sodium bicarbonate is a commonly used product for indigestion treatment. Sodium bicarbonate is a weak base, and the addition of water does not significantly change the pH of the solution. The chemical reaction between sodium bicarbonate and water can be represented by the following equation: NaHCO3(s) + H2O(l) → Na+(aq) + HCO3-(aq) In this reaction, aqueous sodium ions (Na+) and bicarbonate ions (HCO3-) are formed. Let's look at why 90% of the mass of products is not carbon dioxide.When sodium bicarbonate (also known as baking soda) is added to water, it forms a mildly basic solution. If you mean something else, like 90% of the mass of the components will become carbon dioxide, then that probably will not happen. How much is enough acid? We'll get to that in a minute. 90% or more of the bicarbonate ions in sodium bicarbonate will become carbon dioxide), then yes, that will happen if there is enough acid. I'm not sure what you mean by 90% and 10%.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
